Winter 2026
A clear understanding of how to apply appropriate analytical methods is essential to obtain upper-bound estimates of potential patient exposure and to ensure that medical device extractables are fully characterized.
Chemical characterization is critical for understanding potential patient risks associated with exposure to medical device materials. Extractables and potential leachables – substances that are released from medical devices during extraction and clinical use, respectively – are identified during chemical characterization and represent a worst-case exposure assessment for patients using a medical device. The International Organization for Standardization (ISO) 10993-18 standard offers guidelines for best practices to follow for two main tasks required for chemical characterization (ISO, 2020). The first task is to generate one or multiple extract(s) that are suitable both for the duration of use of a particular medical device and for capturing the range of potential chemistries that could be released from the device materials. This task includes handling the extracts appropriately to ensure the analytes contained therein are retained through sample preparation steps (e.g., sample concentration, liquid-liquid extraction).
The second task, which is the focus of this article, involves accurately identifying and quantifying the extractables and leachables contained in the extracts. Table 4 in ISO 10993-18 offers clear instructions based on different analyte characteristics to understand which methods should be applied for specific analyte classes. For example, organic extractables are grouped into three categories defined by their volatilities: volatile organic compounds (VOCs), semi-volatile organic compounds (SVOCs), and non-volatile organic compounds (NVOCs). The volatility of the compound determines which analytical chemistry method is most suitable for its detection.
Extractables and potential leachables are identified during chemical characterization and represent a worst-case exposure assessment for patients using a medical device.”
Two general types of analytical methods, namely chromatography and bulk residue determination, are used for the detection of extractables. In ISO 10993-18, each of the three extractables categories has example methods listed for both types: 1) identification and quantification of individual analytes using chromatographic methods; and 2) quantification of total analytes using bulk residue determination methods. Specifically, chromatographic methods are techniques used to separate, identify, and quantify individual analytes, while bulk residual methods quantify the total amount of analytes.
Chromatography is used to separate the components of a mixture, so that the individual components can be analyzed and identified. Chromatographic methods provide a list of chemical names and the estimated quantity of each chemical extracting from the device, information that is particularly critical for a toxicological risk assessment. A summary of the recommended chromatographic methods for each analyte class is shown in Table 1.

In an extract container, there are separate liquid and gas phases (see Figure). Once generated, VOCs are expected to have a greater affinity for existing in the gas phase above the extract. Therefore, headspace-gas chromatography mass spectrometry (HS-GCMS) has the greatest potential for producing the most accurate quantitation value for VOCs because this method samples directly from the headspace above the extract. Further, gas chromatography (GC) is an alternative option for measuring VOCs; however, since measurements taken using this method are representative of the liquid phase, concentrations of highly volatile compounds could be underestimated. At the same time, GC presents a credible alternative for measuring peripheral VOCs that are on the cusp of being classified as SVOCs.
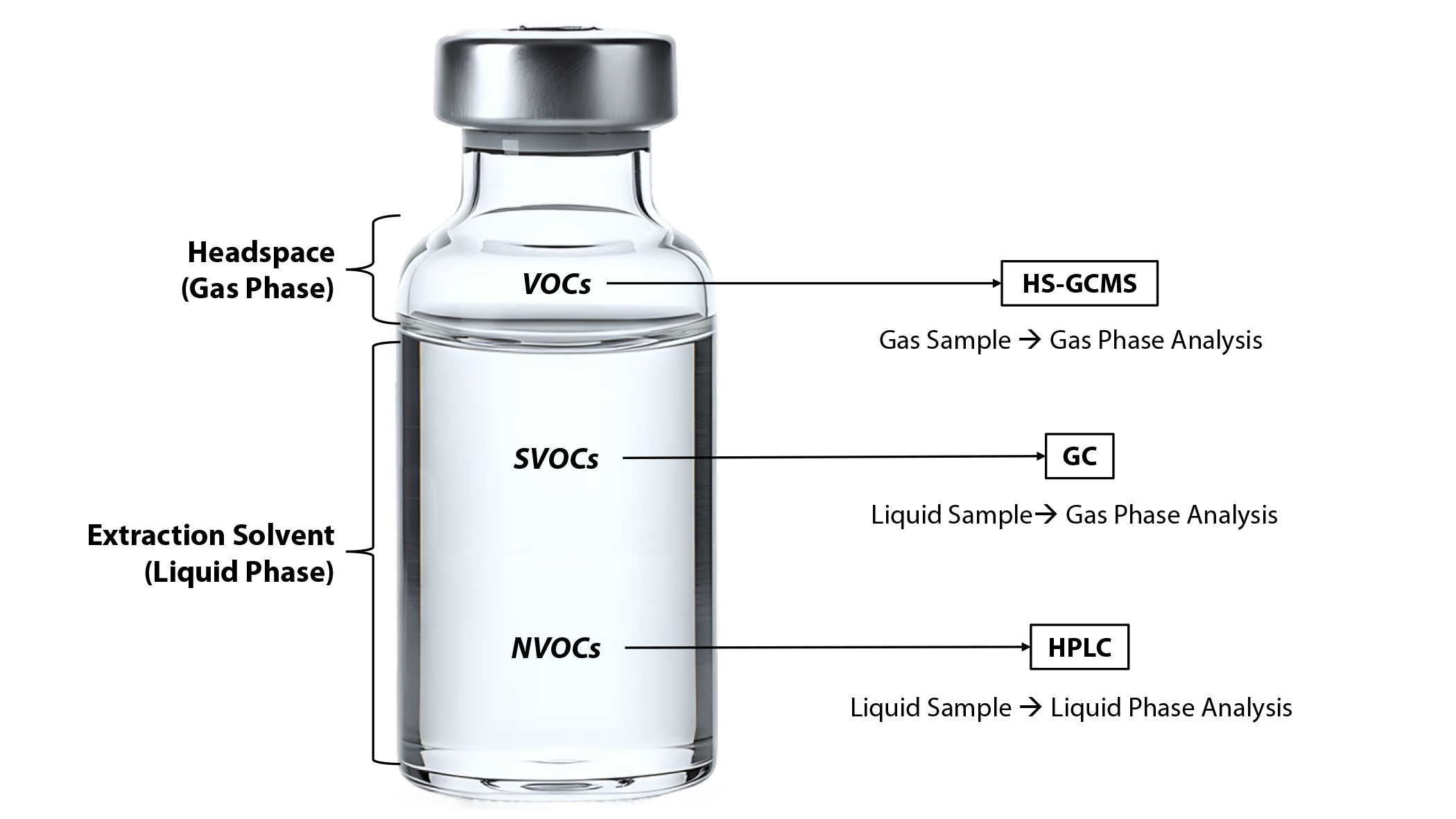
While SVOCs are able to enter the gas phase similar to VOCs, they are expected to have a greater affinity for existing in the liquid phase of the extract. Therefore, GC has an increased likelihood of detecting the most accurate concentration of SVOCs because this measurement is based on the liquid portion of the sample. HS-GCMS is an alternative option for measuring analytes that are considered peripheral SVOCs but are close to the threshold for VOCs, since this method will be compatible with the concentrations that are found in the gas phase. Conversely, high-performance liquid chromatography (HPLC) is a credible alternative for analytes that are considered peripheral SVOCs but are closer to the threshold for NVOCs, as these compounds will exist primarily in the liquid phase.
NVOCs are expected to exist exclusively in the liquid phase of the extract and are not expected to readily volatilize. These characteristics make NVOCs most compatible with HPLC analysis. HPLC analysis starts with a liquid sample, does not require volatilization, and can be used in tandem with ultraviolet-visible (UV) detection, charged aerosol detection (CAD), evaporative light scattering detection (ELSD), and MS. Within the NVOC analyte class, certain structural elements are more compatible with different detectors. For example, structures containing chromophores (e.g., extractables from polyurethane materials) will respond well to UV detection. Since NVOCs do not have a high propensity for entering the gas phase, detection via alternative chromatographic methods, such as HS-GCMS or GC, is not a credible option for this class of analytes. Therefore, HPLC is the only suggested method for identification and quantification of NVOCs.
Bulk residue determination methods are employed for the quantification of the total amount of analytes. While these methods provide a single value quantifying the total quantity of substances extracting from the device, they do not identify the specific compounds. A summary of the recommended bulk residue determination methods for each analyte class is provided in Table 2.
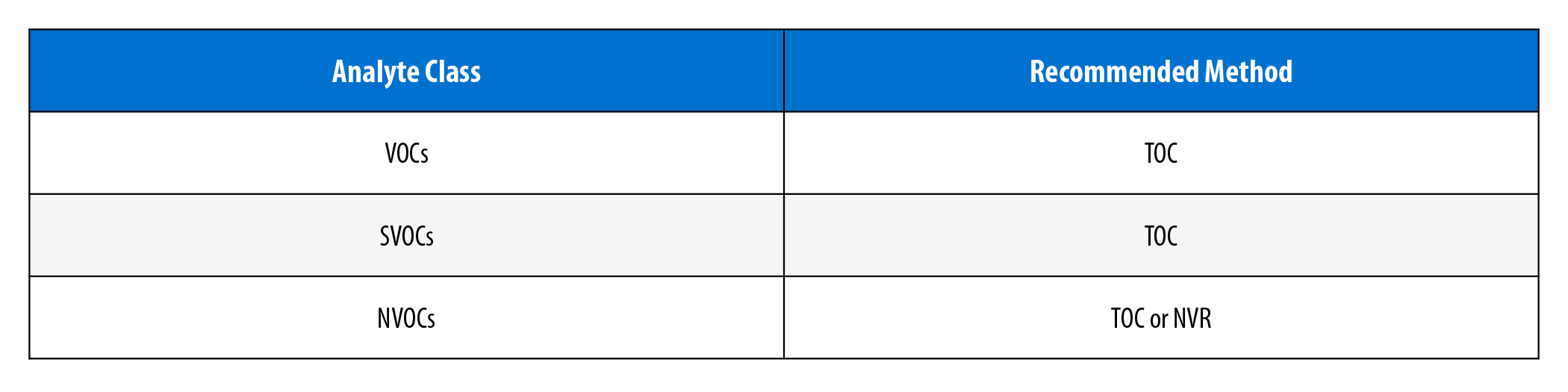
For carbon-containing VOCs, SVOCs, and NVOCs present in aqueous extracts, total organic carbon (TOC) analysis is an appropriate method for determining the amount of organic carbon contained in the residue. Additionally, NVOCs are compatible with non-volatile residue (NVR) determination, an alternative technique that uses gravimetric analysis. The process of gravimetric analysis involves evaporating the solvent from an extract and measuring the mass of the NVR that remains. As previously mentioned, NVOCs are not expected to readily volatilize. Therefore, unlike VOCs and SVOCs, NVR is a credible technique for assessing NVOCs, as this method of residue determination is compatible with organic and aqueous solvents.
Table 4 in ISO 10993-18 is a valuable tool in selecting fit-for-purpose chemical characterization methods. In general, multiple analytical techniques are needed to fully characterize a medical device extract. Understanding how to apply analytical techniques per ISO 10993-18 to detect a wide range of chemicals will result in a more accurate estimate of patient exposure for medical device risk assessments.
The author can be reached at Stephanie.Kearing@gradientcorp.com.
International Organization for Standardization (ISO). 2020. “ISO 10993-18:2020: Biological evaluation of medical devices – Part 18: Chemical characterization of medical device materials within a risk management process (Second Edition).” ISO 10993-18:2020 (E). 74p.