Winter 2026
Cross-referencing various sources of available information regarding extractables and classes of hazardous chemicals offers a valuable screening tool for assessing the potential health risks of medical devices.
Exposures to chemicals released from medical devices during normal use can lead to significant adverse health effects in patients; therefore, chemical characterization of medical device extractable profiles is a critical component of the toxicological risk assessment process required for regulatory approval of medical devices. Identification of all compounds released from the device allows for determination of potential health risks – from systemic toxicity and carcinogenicity to developmental effects and reproductive toxicity – that may result from device use. Recently, several peer-reviewed manuscripts have been published wherein large datasets of medical device extractables have been compiled from chemical characterization studies to provide a comprehensive snapshot of chemicals of concern released from medical devices under clinical use conditions. Two notable examples, Builee et al. (2025) and Borton and Coleman (2025), each compiled data obtained from industry manufacturers, testing laboratories, and toxicology consulting firms to generate unique, consolidated medical device extractables datasets yielding a total of 790 and approximately 18,300 compounds, respectively. To assess potential health hazards, these two lists of organic medical device extractables were compiled and screened for chemicals of concern.
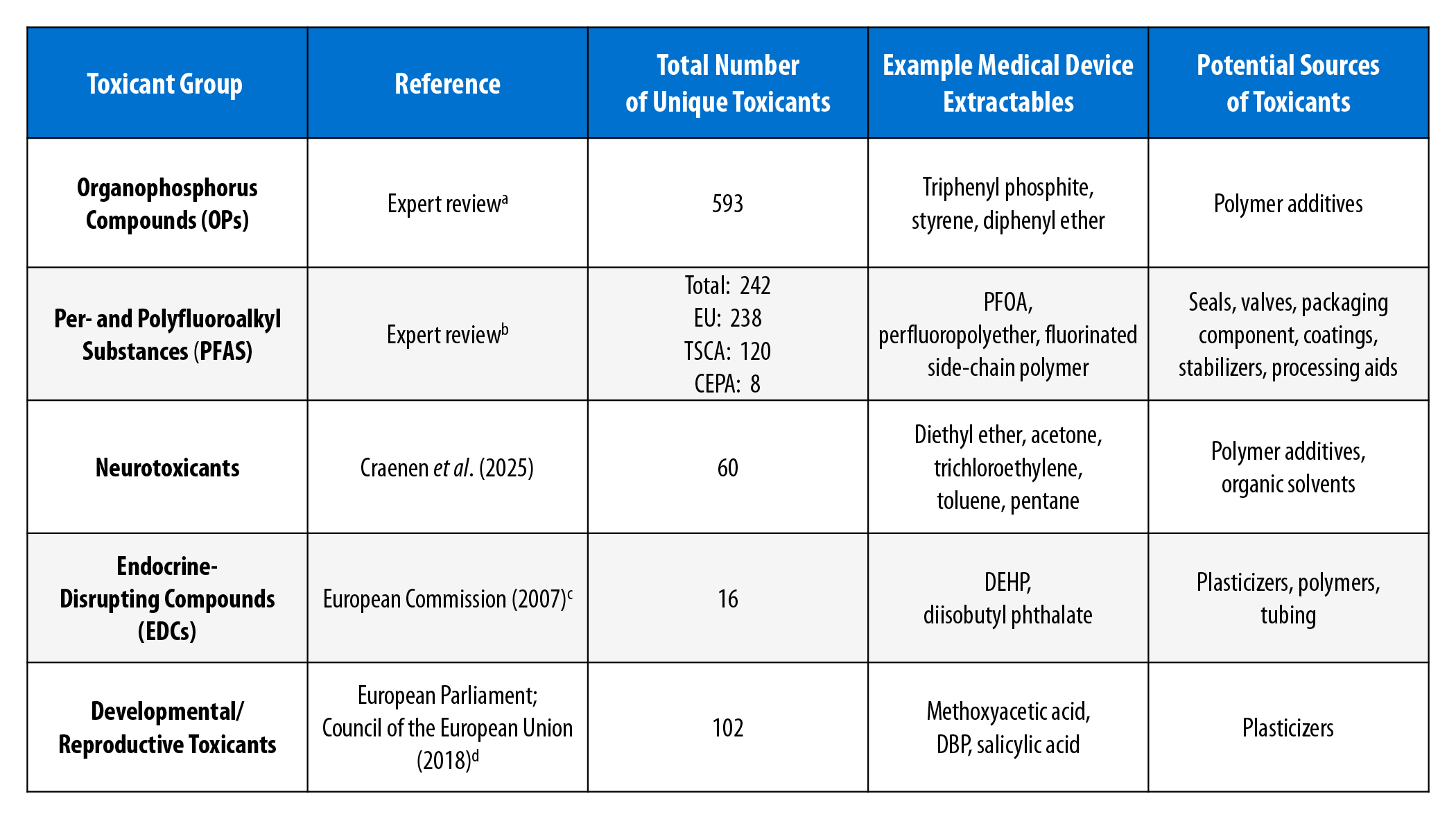
The medical device extractables lists were screened for specific chemicals belonging to the following classes: neurotoxicants, endocrine-disrupting compounds (EDCs), developmental and reproductive toxicants, organophosphorus compounds (OPs), and per- and polyfluoroalkyl substances (PFAS). To conduct the screenings, potential EDCs were identified using the European Chemicals Agency (ECHA) Substances of Very High Concern (SVHC) list; reproductive toxicants were identified from the ECHA Annex VI Classification, Labelling, and Packaging (CLP) list; and neurotoxicants were identified from the reference list of neurotoxic substances based on the hazard conclusions made by the Committee for Risk Assessment (RAC) reported by Craenen et al. (2025). To identify OPs on the medical device extractables datasets, each list was first screened to isolate chemicals that contain one or more carbon-phosphorus bond(s). The list of OPs was then screened against the chemicals used to derive (Munro et al., 1999) and verify (EFSA, 2012) the OP threshold of toxicological concern to assess the overlap between chemical constituents. To identify PFAS, the medical device extractables lists were screened to isolate fluorinated chemicals, and expert review was conducted to classify chemicals as PFAS using various regulatory definitions, including that of the European Union (EU) (ECHA, 2025), the United States (US) Toxic Substances Control Act (TSCA) (US EPA, 2025), and the Canadian Environmental Protection Act (CEPA) list. A summary of the results of this screening is shown in the Table.
Notably, our approach focuses on hazard rather than risk, as the concentration released from medical devices is critical to understanding the likelihood that these devices will elicit a toxicological effect.”
The 60 neurotoxicants identified in the medical device extractables datasets were comprised mostly of organic solvents. These solvents or agents are used typically during manufacturing processes and are likely detected at residual levels on the devices. Further, we identified 16 EDCs and 102 reproductive toxicants. The majority of the reproductive toxicants identified were classified as Category 1B (presumed reproductive toxicant based largely on animal evidence) under ECHA’s CLP Regulations, while only four of the reproductive toxicants were classified as Category 1A (presumed reproductive toxicant based on human evidence). Four OPs were uniquely identified in the medical device extractables lists to be in the Munro and/or the EFSA dataset; 2 of 31 OPs from the Munro dataset and 4 of 51 OPs from the EFSA dataset were on the lists of medical device extractables OPs. These four OPs were all thiophosphate-related compounds.
Of the medical device extractables, 2.6 percent of chemicals were found to be fluorinated and 1.3 percent were identified as unique PFAS. Concordances between the lists of ECHA-, TSCA-, and CEPA-defined PFAS ranged from 53 percent to 75 percent. Additionally, the PFAS were classified further as polymers and non-polymers, according to Henry et al. (2018). Only two of the PFAS were categorized as polymers – neither of which exhibits appropriate characteristics to be classified as a fluoropolymer – and 242 PFAS were classified as non-polymers (Henry et al., 2018). Although only two of the identified PFAS were determined to be polymers, this finding does not reflect a lack of polymers used in medical device manufacturing, as fluorinated-side chain polymers are known to break down into dimers and monomer subunits in biological environments. Upon review of the chemical structures of these compounds, the PFAS identified in the medical device extractables lists likely originated from processing aids, as well as patient-contacting components of the device.
Our analyses confirm that chemicals released from medical devices may include neurotoxicants, developmental and reproductive toxicants, OPs, and PFAS. Notably, our approach focuses on hazard rather than risk, as the concentration released from medical devices is critical to understanding the likelihood that these devices will elicit a toxicological effect. By evaluating chemicals of concern (e.g., EDCs, cohorts of concern) known to be released from medical devices, medical device manufacturers can select safer materials and manufacturing processes. Moreover, integrating large-scale medical device extractable datasets with compiled lists of known toxicants of different hazard classes provides a robust framework for identifying and managing the potential risks posed by chemical hazards released from medical devices. This approach, paired with cutting-edge biological evaluations, can help to improve confidence in the non-targeted screening analysis of medical device extractables, anticipate regulatory changes, and guide medical device manufacturers toward safer product designs, ultimately improving patient safety and public health.
The authors can be reached at. Katherine.Roach@gradientcorp.com and Brigitte.Cronin@gradientcorp.com.
Borton, L; Coleman, K. 2025. “Material-mediated pyrogens in medical devices: Myth or reality?” ALTEX doi: 10.14573/altex.2504231.
Builee, T; Kennedy, TA; Levelut, V; Hahn, MA; Bond, S; Peterson, MK; Hsia, FK; Stornetta, A; Erickson, KJ; Ehman, KD; Prabhakar, B; Bagley, BD; Parker, SP. 2025. “Characterization of medical device constituents and development of duration-based non-cancer threshold of toxicological concern values.” Front Toxicol. 4(7):1600127. doi: 10.3389/ftox.2025.1600127.
Craenen, K; Kosiaras, P; Hellsten, K. 2025. “A reference list of neurotoxicants based on CLP harmonised classifications.” Neurotoxicology 109:11-26. doi: 10.1016/j.neuro.2025.05.006.
European Chemicals Agency (ECHA); Committee for Risk Assessment (RAC), Committee for Socio-Economic Analysis (SEAC). 2025. “Background document to the Opinion on the Annex XV dossier proposing restrictions on Per- and polyfluoroalkyl substances (PFASs) (Draft Version 14).” June 24. Accessed on November 6, 2025, at https://echa.europa.eu/hot-topics/perfluoroalkyl-chemicals-pfas#portlet_com_liferay_journal_content_web_portlet_JournalContentPortlet_INSTANCE_dvljVDl4JyqT.
European Commission. 2007. “Corrigendum to Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), establishing a European Chemicals Agency, amending Directive 1999/45/EC and repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94 as well as Council Directive 76/769/EEC and Commission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC (Official Journal of the European Union L 396 of 30 December 2006).” Off. J. Eur. Union L 136:3-280, May 29.
European Food Safety Authority (EFSA). 2012. “Scientific opinion on exploring options for providing advice about possible human health risks based on the concept of threshold of toxicological concern (TTC).” EFSA J. 10(7):2750.
European Parliament; Council of the European Union. 2018. “Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006 (Text with EEA relevance).” 1389p, March 1. Accessed on March 11, 2019, at https://eur-lex.europa.eu/legal-content/en/TXT/PDF/?uri=CELEX:02008R1272-20180301&from=EN.
Henry, BJ; Carlin, JP; Hammerschmidt, JA; Buck, RC; Buxton, LW; Fiedler, H; Seed, J; Hernandez, O. 2018. “A critical review of the application of polymer of low concern and regulatory criteria to fluoropolymers.” Integr. Environ. Assess. Manag. 14(3):316-334. doi: 10.1002/ieam.4035.
Munro, IC; Kennepohl, E; Kroes, R. 1999. “A procedure for the safety evaluation of flavouring substances.” Food Chem. Toxicol. 37(2-3):207-232. doi.org: 10.1016/S0278-6915(98)00112-4.
US EPA. 2025. “Perfluoroalkyl and polyfluoroalkyl substances (PFAS) data reporting and recordkeeping under the Toxic Substances Control Act (TSCA); Revision to regulation (Proposed rule).” Fed. Reg. 90(217):50923-50937. 40 CFR 705, November 13. Accessed on November 14, 2025, at https://www.govinfo.gov/content/pkg/FR-2025-11-13/pdf/2025-19882.pdf.